Modern (1940’s-present)
73 The COVID-19 Vaccine
Kevin Dai
Introduction

In December 2019, the new coronavirus SARS-CoV-2, shown in Fig.1, gave rise to the COVID-19 pandemic. The virus, which was first observed to be causing clusters of pneumonia cases, rapidly spread worldwide, leading the World Health Organization (WHO) to declare a pandemic on March 11, 2020, and a Public Health Emergency of International Concern on January 30, 2020. The pandemic’s unparalleled scope and consequences have caused extensive disruptions to healthcare systems, the economy, and everyday life globally, with efforts to contain its spread persisting into the following years.
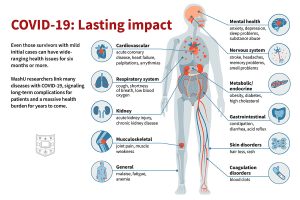
About a year later, the development and widespread distribution of the COVID-19 vaccines have given hope to combat a global pandemic caused by the new SARSCo2V. Although these vaccines have been shown to reduce virus spread and severity, there are concerns about possible short and longer-term adverse reactions, as depicted in Fig.2. Due to numerous accounts of uncommon but severe side effects following vaccination, there is widespread worry regarding their safety. Myocarditis, lymphadenopathy, thrombocytopenia, pulmonary embolism, and myocardial infarction are among these side effects. Understanding the short and long-term impacts of COVID-19 vaccinations is crucial for making well-informed decisions and managing public health effectively. In the short term, individuals may experience mild to moderate side effects, such as pain at the injection site, fatigue, headache, and fever, which indicate the body’s immune response. However, as more data are collected in post-marketing surveillance studies and clinical trials, the evaluation of long-term side effects requires continuous monitoring and further research. While the available data indicates that severe long-term side effects from COVID-19 vaccinations are uncommon, the general public still deserves a comprehensive explanation and improved understanding of the relevant research findings. To protect individuals and communities from COVID-19, it is important to balance the need to address concerns and to emphasize the benefits of vaccination.

COVID-19 Vaccine
COVID-19 vaccines represent a monumental breakthrough in the global fight against the pandemic caused by the SARS-CoV-2 virus. Developed through rigorous research and clinical trials, these vaccines provide a crucial defense mechanism against COVID-19 infection by stimulating the body’s immune response to recognize and combat the virus. By triggering the production of antibodies, they significantly reduce the risk of severe illness, hospitalization, and death from COVID-19. With various types of vaccines available, including mRNA, viral vector, and protein subunit vaccines, efforts to vaccinate populations worldwide have been underway to curb the spread of the virus and achieve herd immunity. However, continued vigilance in monitoring vaccine efficacy, safety, development, and distribution remains essential to ensure widespread protection and bring an end to the global health crisis. Science and technology are intricately connected to the development, manufacturing, distribution, and monitoring of COVID-19 vaccines. Scientists conduct comprehensive clinical trials to ensure the vaccines’ safety, while following large-scale manufacturing and meticulous cold chain management highlight the immense impact of science and technology on our society’s ability to combat the pandemic.
Types of COVID-19 Vaccines
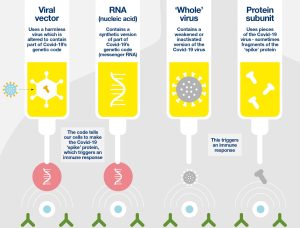
Several brands of COVID-19 vaccines have emerged as key players in the global vaccination campaign. Different types of COVID-19 vaccines works differently, as presented in Fig.3. The Pfizer-BioNTech and Moderna vaccines are mRNA-based vaccines that utilize genetic material from the virus to stimulate an immune response. Similarly, the Johnson & Johnson and AstraZeneca vaccines employ viral vector technology, where a harmless virus delivers genetic instructions to cells to produce viral proteins, triggering an immune response. Other vaccines, such as Sinovac and Sinopharm, are inactivated virus vaccines, containing killed versions of the virus to elicit an immune response. Each vaccine brand has undergone rigorous testing to ensure safety and efficacy, with varying dosing schedules and storage requirements. Despite differences in technology and administration, all authorized vaccines play a critical role in the global effort to control the spread of COVID-19 and bring an end to the pandemic. (ChatGPT, personal communication, April 10, 2024)
Vaccine Hesitancy
Effective communication of scientific knowledge is critical for building public trust and increasing vaccine uptake. Technology act as an important role in conveying correct information via digital platforms, social media, and telecommunication channels, while scientific research influences message strategies and public health initiatives. Since the release of the COVID-19 vaccine, many people have started to ask questions regarding how quickly it was developed. There are doubts on internet as to whether the vaccines have undergone sufficient clinical testing to prove they are safe for general usage. Before we jump into the studies regarding the side effects of the COVID-19 vaccine, let’s take a moment to understand how cultural differences also can play a critical role in vaccine hesitancy.
The term “COVID-19 vaccine hesitancy” particularly describes people who, although the vaccination is available, are reluctant or refuse to receive it. Like vaccine hesitancy in general, COVID-19 vaccine reluctance can be brought on by personal beliefs and values, misinformation circulating on social media or other sources, mistrust of pharmaceutical corporations or the healthcare system, and worries about the safety and effectiveness of the vaccinations. The global effort to contain the COVID-19 pandemic is crucial because achieving herd immunity and stopping the virus’s spread need mass vaccination. To overcome COVID-19 vaccine reluctance, effective communication plans, community outreach initiatives, and open distribution of factual information regarding the vaccine’s safety, effectiveness, and significance in containing the pandemic are all necessary.
Lu’s study mapped COVID-19 vaccination hesitancy globally by analyzing data from daily surveys of around 12 million people in 244 countries. It was shown that there was a correlation between higher levels of vaccine hesitancy and specific demographic and contextual characteristics. In particular, vaccine hesitancy was more common in people who were female, younger, less educated, or lived in nations with laxer government response times (Lu, 2023). The study also examined the psychological and cultural facets of vaccine hesitancy, and it was found that the cultural factor of uncertainty avoidance was important. Initially, in late 2020, there was a greater level of hesitancy due to concerns about vaccine adverse effects among persons in nations with higher uncertainty avoidance. However, these disparities decreased with time as more people received the COVID-19 vaccination (Lu, 2023).
The Short-term Side Effect
The term “short-term side effects” refers to COVID-19 vaccine side effects that usually manifest a few days following the vaccination. These side effects can last up to a week and often appear one to three days after vaccination.
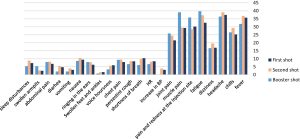
In Haider’s Pfizer vaccination research, he comprised 1000 participants in the Pakistani population, 64.4% male and 35.6% female, with an average age of 43.06±14.98 years. Among them, Haider included 280 people with hypertension and 356 people with diabetes in the survey to provide a more realistic picture of the health of society. This strategy aimed to take account of various potential causes and minimize the uncertainties within the sample population. After the initial injection of the first dose of the Pfizer vaccine, the most common side effects reported were burning at the injection site (70.4%) and fever (70.0%), which were then followed by injection site discomfort (63.2%) and muscular soreness (63.2%). Swelling (52.8%), joint discomfort (58.0%), sore throat (52.8%), and chills (46.0%) were additional adverse symptoms. The least amount of reports (20.0% and 18.0%, respectively) included nausea and redness at the injection site. All the side effects are summarized within the Fig.4 bar graph. Following the second dosage, the most commonly reported side effects were muscle pain (62.8%), rashes (60.8%), and injection site pain (57.6%) (Haider et al., 2023). In addition to Haider’s research, Khan’s research especially sought to investigate the short-term adverse effects in both healthy individuals and diabetic subjects. The frequency of diabetes underscores the relevance of comprehending how immunization affects this population as a comorbidity among COVID-19 patients. His studies reveal that COVID-19 vaccination side effects are more common in diabetic patients than in non-diabetic ones (Khan et al., 2023). The most frequently reported side effects among participants with diabetes were edema at the injection site, fever, burning feeling at the injection site, and muscle and joint pain. To ensure the safety and well-being of diabetic patients receiving the COVID-19 vaccine, he recommends and highlights the necessity for healthcare practitioners to monitor and treat any adverse responses properly (Khan et al., 2023).
Meanwhile, Abukahlil’s Pfizer vaccination study included 1137 participants from Birzeit University in Palestine with a mean age of 21.163 ± 5.361 years. Among the participants, 66.8% were female, 91.8% were in good health with no chronic illnesses, 26.7% smoked, 14.4% reported dietary or drug allergies, and 63.2% were older than 20 years old. In contrast to Haider’s research, this study clearly focused on a younger generation with a mean age difference of around 22 years lower. Based on the study findings, the most often reported side effects included fever, chills, headache, exhaustion, injection site pain and edema, muscular soreness, and joint pain, as shown in Fig 2. “Rare and severe side effects that require medical attention were reported by eight participants, including one (0.1%) case of myocarditis and 7 (0.6%) cases of thrombocytopenia” (Abukhalil et al., 2023). More research is still required to determine the relationship between the small percentage of people who experience severe side effects and their causation.
When all the above COVID-19 immunization cases are considered, it becomes clear that fever, chills, headaches, fatigue, injection site discomfort, joint pain, and muscle soreness are the most often reported short-term adverse effects. The short-term side effects pose no significant danger, and there is little need for excessive concern. In the majority of cases, they will resolve on their own within approximately a week.
The Long-term Side Effect
“Long-term side effects” refer to COVID-19 vaccine side effects that appear or worsen several weeks, months, or even years after the vaccination. Since the long-term side effects of the COVID-19 vaccination are less common than the short-term side effects, more studies must be done over a more extended period of time in order to understand the impact and prevalence of these side effects fully.
Concerns about the safety of COVID-19 vaccinations and their impact on platelet function are widespread. According to Professor Zhou, she believes there is a connection between the COVID-19 vaccine and platelet aggregate concentrations. When the platelet aggregate concentration is at a certain level, unusual blood clotting will happen, which increases the risk of stroke and heart attacks. In her research, the concentration of platelet aggregates is defined by the proportion of images obtained with platelet aggregates to all images obtained for every blood sample; this ratio acts as a direct measure of platelet activity (Zhou et al., 2022). Over the course of around a year, participants got several doses of the Pfizer-BioNTech (BNT162b2) vaccine. Following the vaccine doses, there are no discernible or long-lasting patterns of platelet aggregation in Professor Zhou’s experimental data. This implies that vaccinations had a very minimal impact on platelet activation, turnover, aggregation, and leukocyte interaction, which resolves one of the primary mysteries and concerns among the public regarding the potential long-term negative effects of the COVID-19 vaccine.
Hosseini and Askari have provided a comprehensive review of documents reporting neurological side effects of COVID-19 vaccines in international databases from 2020 to 2022 and discussed neurological disorders possibly caused by vaccination. According to their findings, COVID-19 vaccination side effects are more common in those with a history of immune-related disorders or in people who are more vulnerable because of their age or physical characteristics (Hosseini & Askari, 2023). Cerebral venous sinus thrombosis, connected explicitly to the AstraZeneca vaccine, and transverse myelitis, associated with the Pfizer, Moderna, AstraZeneca, and Johnson & Johnson vaccines, are among the most serious and often reported side effects. Furthermore, the Pfizer, Moderna, and AstraZeneca vaccines have been associated with a higher incidence of Bell’s palsy, and the Pfizer, AstraZeneca, and Johnson & Johnson vaccines have been associated with a higher incidence of Guillain-Barré syndrome (GBS). Additionally, there have been cases of multiple sclerosis (MS) with no prior symptoms, most notably related to the Pfizer vaccination (Hosseini & Askari, 2023).
Nevertheless, clear evidence of causality will require more research, continuous data gathering, and long-term monitoring to determine whether these problems are accidental or directly induced by the vaccine. These results highlight the significance of ongoing monitoring and careful investigation of adverse events related to vaccinations to guarantee the security and effectiveness of COVID-19 immunization programs.
Conclusion
To conclude, the global response to the COVID-19 pandemic has made significant progress in developing and deploying vaccinations. These vaccines promise to slow the virus’s transmission and minimize its effects on public health and society. However, worries about vaccine safety and potential side effects remain, particularly short and long-term adverse reactions. Research studies have provided important insights into the nature and occurrence of these side effects, which range from moderate to severe. Short-term side effects include fever, tiredness, and injection site soreness, which usually pass within a week. However, worries about rare but major adverse effects, including as neurological diseases and blood clotting abnormalities, demand continued monitoring and investigation. Cultural and psychological variables and individual health situations contribute to vaccine reluctance, emphasizing the significance of targeted communication and outreach initiatives to address public concerns and promote broad vaccination. While evidence suggests that long-term severe side effects from COVID-19 vaccinations are uncommon, further vigilance and study are required to assess their safety and effectiveness fully.
Navigating the complexity of the pandemic and protecting public health requires a balanced approach that considers both the benefits and potential risks of vaccination. Through sustained collaboration and effort, we can work together to overcome the obstacles posed by COVID-19 and usher in a brighter, healthier future for all individuals.
Chapter Activity
1. Name three different types of COVID-19 vaccine and briefly explain how the vaccine works in a few sentences.
2. The development of the COVID-19 vaccine occurred at a similar pace compared to traditional vaccine timelines, which typically span several years or even decades. (True or False)
References
Abukhalil, A. D., Shatat, S. S., Abushehadeh, R. R., Al-Shami, N. M., Naseef, H. A., & Rabba, A. (2023). Side effects of Pfizer/BioNTech (BNT162b2) COVID-19 vaccine reported by the Birzeit University community. BMC Infectious Diseases, 23(1), 5. https://link.springer.com/article/10.1186/s12879-022-07974-3
Haider, S. M. S., Alvi, S. A., Khan, H., Majeed, R., Syed, T., Anwar, A., & Hashmi, A. A. (2023). Common Side Effects of Pfizer COVID-19 Vaccine: An Experience From Pakistan. Cureus, 15(6). https://pubmed.ncbi.nlm.nih.gov/37492805/
Hosseini, R., & Askari, N. (2023). A review of neurological side effects of COVID-19 vaccination. European Journal of Medical Research, 28(1), 1-8. https://eurjmedres.biomedcentral.com/articles/10.1186/s40001-023-00992-0?s=09
Khan, F., Khan, M. T., Zaman, S., Mujtaba, S., Batool, A., Ghanghro, Z., … & Hashmi, A. A. (2023). Side effects of COVID-19 vaccines among diabetic subjects and healthy individuals. Cureus, 15(3). https://assets.cureus.com/uploads/original_article/pdf/143361/20230410-17889-o13j61.pdf
Lu, J. G. (2023). Two large-scale global studies on COVID-19 vaccine hesitancy over time: Culture, uncertainty avoidance, and vaccine side-effect concerns. Journal of Personality and Social Psychology, 124(4), 683–706. https://doi.org/10.1037/pspa0000320
Zhou, Y., Nishikawa, M., Kanno, H., Yang, R., Ibayashi, Y., Xiao, T., Peterson, W., Herbig, M., Nitta, N., Miyata, S., Kanthi, Y., Rohde, G. K., Moriya, K., Yatomi, Y., & Goda, K. (2022). Long‐term effects of Pfizer‐BioNTech COVID‐19 vaccinations on platelets. Cytometry Part A, 103(2), 162–167. https://doi.org/10.1002/cyto.a.24677
Figures
“Among COVID-19 survivors, an increased risk of death, serious illness” by Julia Evangelou Strait, Washington University School of Medicine in St. Louis is licensed under CC BY-NC 4.0.
“Coronavirus disease (COVID-19) is an infectious disease caused by the SARS-CoV-2 virus” by World Health Organization (WHO) is licensed under CC BY-NC 4.0
“Possible Side Effects Of COVID-19 VACCINE” by Bangkok Hospital is is licensed under CC BY-NC 4.0.
“Side effects of Pfizer/BioNTech (BNT162b2) COVID-19 vaccine” by Abukhalil et al. is licensed under CC BY-NC 4.0.
“What different types of Covid-19 vaccine are there?” by Wellcome is licensed under CC BY-NC 4.0.
AI Acknowldegement
I acknowledge the use of ChatGPT to gather resources regarding the potential side effects of COVID-19 vaccine. The prompts used include “COVID-19 vaccine”, “different types of COVID-19 vaccine”, “short-term and long-term side effects of Pfizer Covid vaccine,” “potential side effect of Covid vaccine” and more. The output from these prompts was used to brainstorm ideas, form a general outline, write paragraphs, and check the grammar of the written paragraphs.


